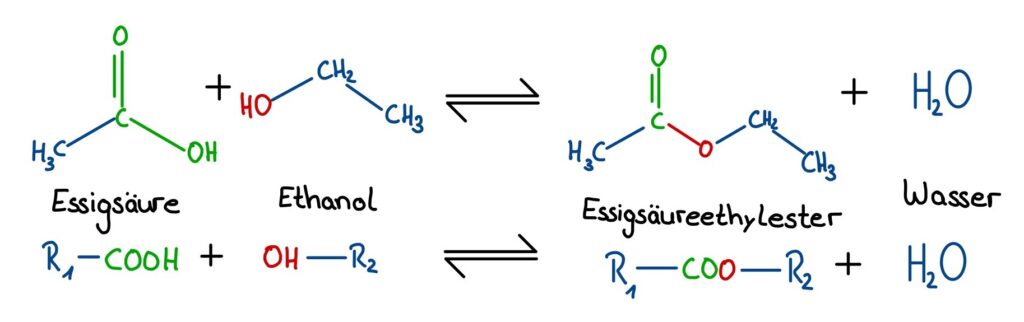
Esters are organic compounds that play an important role in creams and many other areas. In creams, they can serve as preservatives, emulsifiers, or fragrances. Esters are formed through a reaction called esterification. Here, acids react with alcohol with the elimination of water.
The difference between conventional and sustainable production can be clearly illustrated using the example of ester production for a cream. The factors of energy consumption, raw material selection, and the design of the process steps play a crucial role.
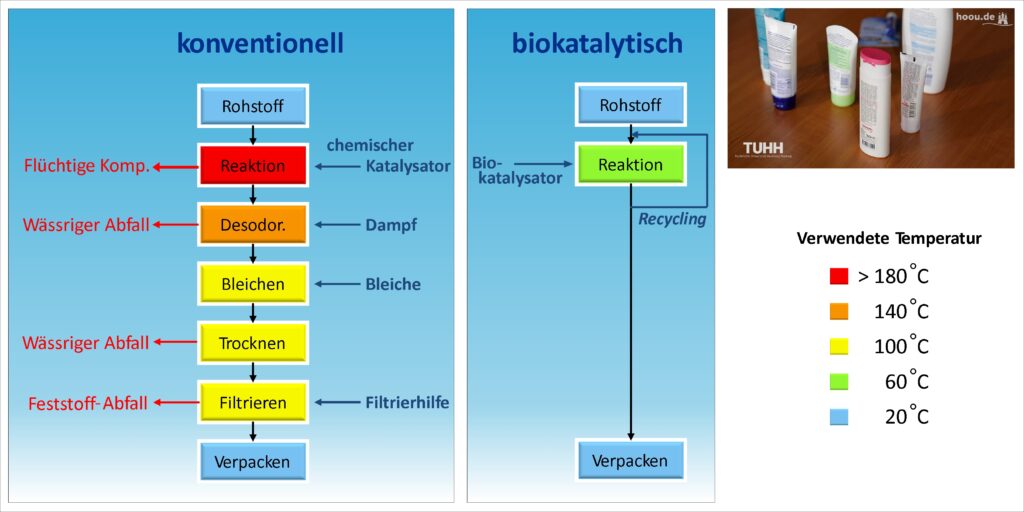
Conventional Process
Reaction
Esterification is carried out with a chemical catalyst (e.g., sulfuric acid), and volatile components are already produced in this step, which must be captured and disposed of. The reaction of the raw materials to the product takes place at over 180 °C and is therefore very energy-intensive.
Deodorization
In deodorization, the product is freed from odors that should not be present in the final product. This step is carried out with steam and is therefore associated with high energy consumption, although the temperature is slightly lower than in the first step. Heating one liter of water to 100 °C requires 418.2 kJ of energy. However, due to the high heat of vaporization of water, an additional 2257 kJ of energy is needed to generate steam from boiling water. This is more than five times the amount of energy required to heat the water to 100 °C. The steam absorbs odors but must then be condensed and disposed of as aqueous waste. (Ideally, some of the energy used can be recovered during condensation.)
Bleaching
Bleaching agents are used to achieve the desired color of the product and to destroy unwanted colorants.
Drying
To obtain the final product, water must be separated from the mixture. The purification of this aqueous waste is expensive, and therefore so is its disposal. As explained in the deodorization process, evaporating water requires a lot of energy.
Filtration
To remove any remaining impurities or particles, the product is filtered. The solids removed during this process are disposed of as waste.
Therefore, five process steps are necessary to obtain the ester usable in the cream through the conventional reaction. The disadvantages are that the reaction must be carried out at high temperatures, steam is required for deodorization, and waste is generated in four process steps that must be disposed of.
Biocatalytic Process
Reaction
Esterification is carried out using an enzyme as a catalyst and can therefore be performed at a significantly lower temperature of 60 °C. All substances that could not be converted in the reaction are fed back into the reaction. This recycling requires fewer raw materials, as none of them are lost as waste. Furthermore, the enzymatically catalyzed reaction does not produce any unwanted by-products that would need to be removed from the product to meet the requirements.
One step? Yes, exactly. The sustainable process requires only one step to produce the final product from the raw materials. The temperature is only 60 °C and no steam is used, which requires much less energy.
In this video, Steffi shows how Claudia runs such a reaction in the bubble column in our labs.
We will go into the bubble column in more detail later. An experiment on building your own bubble column is also offered. So stay tuned!
We have repeatedly mentioned enzymes. But what are enzymes and what are they used for in everyday life? If you want to find out, feel free to check out the following pages about enzymes.
