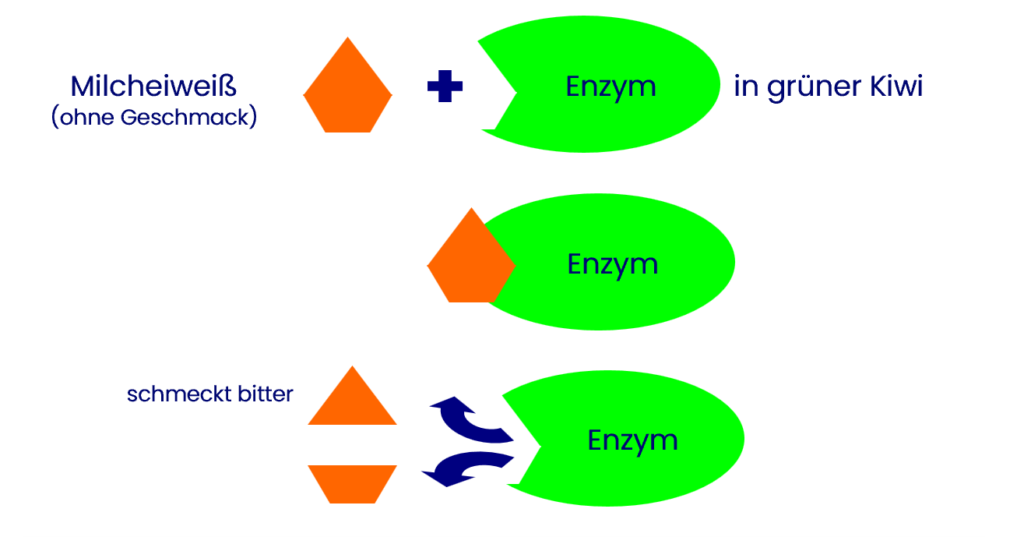
Green Kiwis contain the enzyme actinidin, which breaks down proteins, such as the proteins found in dairy products. Some of the resulting products taste bitter[1], which depends, among other things, on the size of the fragments and their spatial arrangement. Therefore, fresh green kiwis should not be added to desserts or shakes with milk, or should be consumed before the enzyme has produced too many bitter substances.
Yellow Kiwis do not contain this enzyme at all, or it has such low activity that these kiwis can be consumed raw with dairy products without a bitter taste developing[2].
Besides kiwis, intolerance with milk can also be observed with pineapple and papaya, as these tropical fruits also contain many enzymes that split proteins. Depending on the fruit, a different enzyme dominates: actinidin in green kiwi, bromelain in pineapple, and papain in papaya. The fact that enzymes are destroyed when they get too hot can be used to use these fruits with milk anyway. If you pour boiling water over kiwis, pineapples, and papayas before using them and let them steep briefly, the enzymes will lose their effect!
Tip: By the way, enzymes in these types of fruit also bother gelatin! They can prevent gelatin from setting or dissolve already set gelatin. The observations from your experiment help here, because here too, the enzymes in the fruit can be destroyed by heat by pouring the gelatin over these fruits as hot as possible, and the gelatin will set and remain set!
[1] Source:Wieser, H., Belitz, HD. Zusammenhänge zwischen Struktur und Bittergeschmack bei Aminosäuren und Peptiden II. Peptide und Peptidderivate. Z Lebensm Unters Forch 160, 383–392 (1976). https://doi.org/10.1007/BF01106329
[2] Source: Kaur L, Mao B, Bailly J, Oladeji O, Blatchford P, McNabb WC. Actinidin in Green and SunGold Kiwifruit Improves Digestion of Alternative Proteins-An In Vitro Investigation. Foods. 2022 Sep 6;11(18):2739. doi: 10.3390/foods11182739. PMID: 36140865; PMCID: PMC9497782.
And what does this have to do with bioengineering?
Enzymes are nature’s tools for chemical transformations, so-called biocatalysts. They accelerate reactions at mild temperatures where living organisms exist and cause fewer by-products than chemical reactions. Therefore, they are also used in industry.
Bioengineers investigate how such processes can be carried out. Since enzymes are sensitive, it must be investigated under which reaction conditions (e.g., regarding temperature, pH value) the highest possible yield is achieved and the enzymes remain stable and functional for a long time. The origin of the enzymes also plays a role. If such enzymes exist, for example, in bacteria that live in hot springs, then they can tolerate higher temperatures. Further tasks for bioengineers include determining the selection and size of the bioreactor (vessel) in which the transformation takes place. Are stirrers needed to ensure good mass transfer? Which stirring geometry works best? What needs to be considered during “upscaling” when production is to be switched from a small laboratory scale to large industrial processes? How is quality analyzed and ensured? These are some of the questions that bioengineers encounter during their studies and in their profession. You will learn more about this soon in our new biotechnology mission.

(C) TU Hamburg, Institute for Technical Biocatalysis
