
Hello everyone!
That was a bit disappointing! Either the gels melted in the oven or they shrunk. The fruit and vegetables also looked shriveled afterwards. In the picture below you can see our results.
Of course, you can try to see if drying in the air works better – but I don’t have much hope for that! If you want to know why, take a look at the excursion on capillary forces.
Samuel Kistler didn’t have much luck at first either, but he had a laboratory where he had other options available than in a normal kitchen…
So, we’ll now tell you how researchers produce aerogels:
What process steps are necessary for production?
Several process steps are necessary for aerogel production, leading from the hydrogel via the alcogel to the final product aerogel. – But wait a minute: What do these words mean?

- “Hydro” means water, so it’s about a gel in which water is bound in a solid.
- “Alko” stands for alcohol. So, you first pour concentrated alcohol onto the hydrogel, largely replacing the water with alcohol.
- “Aero” means air. At the end of the process, the liquid in the gel is replaced by air.
- “Xero” means “dry”. “Xerogel” is the name for a failed aerogel, i.e., a gel that has shrunk during its production.
Here are the process steps shown schematically:
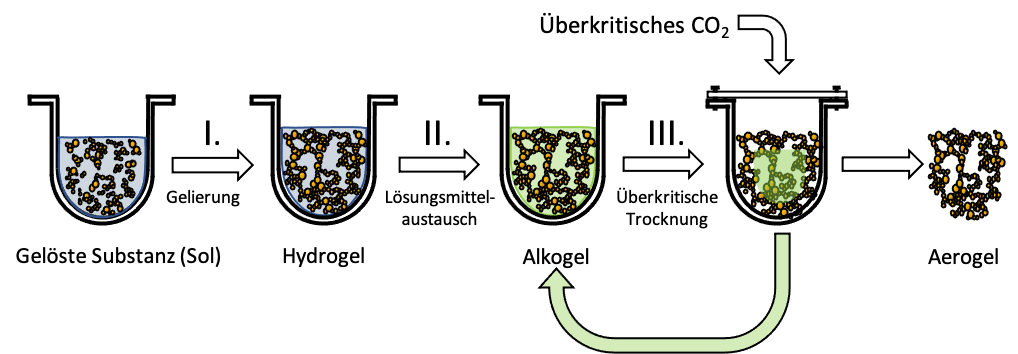
- Gelation: The gel forms from a liquid; the product is a hydrogel.
- Solvent exchange: Water in the pores is replaced by alcohol. The product is an alcogel.
- Supercritical drying: Alcohol in the pores is replaced by air. The product is an aerogel.
Since each intermediate step is important for aerogel production, we will describe each one individually and in detail below. Before that, however, we will briefly discuss the raw materials from which aerogels can be produced.
