Manufacturing Step 2: Solvent Exchange
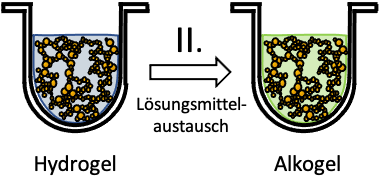
The second step is a preparatory step for the subsequent drying: the hydrogel is converted into an alcogel by replacing the water in the pores with alcohol (chemical name: ethanol). To do this, the gels are immersed in ethanol. The water gradually migrates (diffuses) out of the pores of the gel, while the ethanol penetrates the pores. Ethanol and water mix in the process. Therefore, this liquid must be decanted several times and replaced with fresh ethanol until the gel / fruit has an ethanol content of 97% or higher. This is important for the subsequent drying. Usually, the hydrogels become firmer and less wobbly on their way to becoming alcogels, sometimes they also shrink a little.
Interactive Video:
But why is solvent exchange needed?
Simply put: Because alcohol has a lower boiling point than water and is therefore easier to remove from the pores in the next step and under gentler conditions than water. – Although it’s not quite that simple. For this, we need to explain a little more about states of matter, phase diagrams, and miscibility of substances. You will find these explanations in the excursions “Solvent Exchange” and “Phase Diagram and Supercritical State“. (If you ever study process engineering or chemistry, you will learn this knowledge in the subject “Thermodynamics”.)
