Manufacturing Step 3: Supercritical Drying
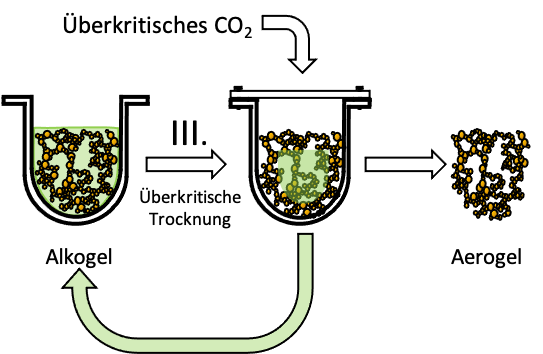
To turn the alcogel into an aerogel, the liquid must be removed from it, meaning the gel must be dried. This drying process is the final manufacturing step. However, it must be a special drying process, because in the experiments in Mission 2b you saw that the gel shrinks during normal drying and the structure collapses.
The goal of aerogel manufacturing is, however, to preserve the structure that formed during gelation, meaning to ensure that the water in the pores is replaced by air without damaging this fine framework.
Why can’t the water simply be removed from the pores by evaporation?
The answer lies in the pore structure of the hydrogel. If the water (or another liquid) evaporates in the pores, so-called capillary forces act, which pull the inside of the pore together and thus destroy the framework. The gel would shrink. To avoid shrinkage due to capillary forces, the gel must not notice that the liquid is being replaced by gas. This is where supercritical drying comes into play: it utilizes the fact that by increasing pressure and temperature, the so-called supercritical state of a substance can be reached, in which there is no distinction between gas and liquid phases. The supercritical state of a substance occurs as soon as the critical point is exceeded (31°C and 74 bar for CO2).
Such high pressure can only be generated in a gas-tight container that can withstand high pressures. This is called an autoclave. The principle is like a pressure cooker, only that even higher pressures (over 100 bar!) can be reached in an autoclave.
Interactive Video:
During supercritical drying, an aerogel is produced from an alcogel by flushing with supercritical carbon dioxide, whose pores initially contain carbon dioxide instead of solvents, which is gradually replaced by air after removal from the autoclave. Since the pores and the shape of the gel are preserved during supercritical drying, the gel does not shrink, so the volume remains the same. However, the weight decreases simultaneously, as air is much lighter than the liquid that was previously in the pores. Therefore, dry aerogels are extremely light and have a very low density.
